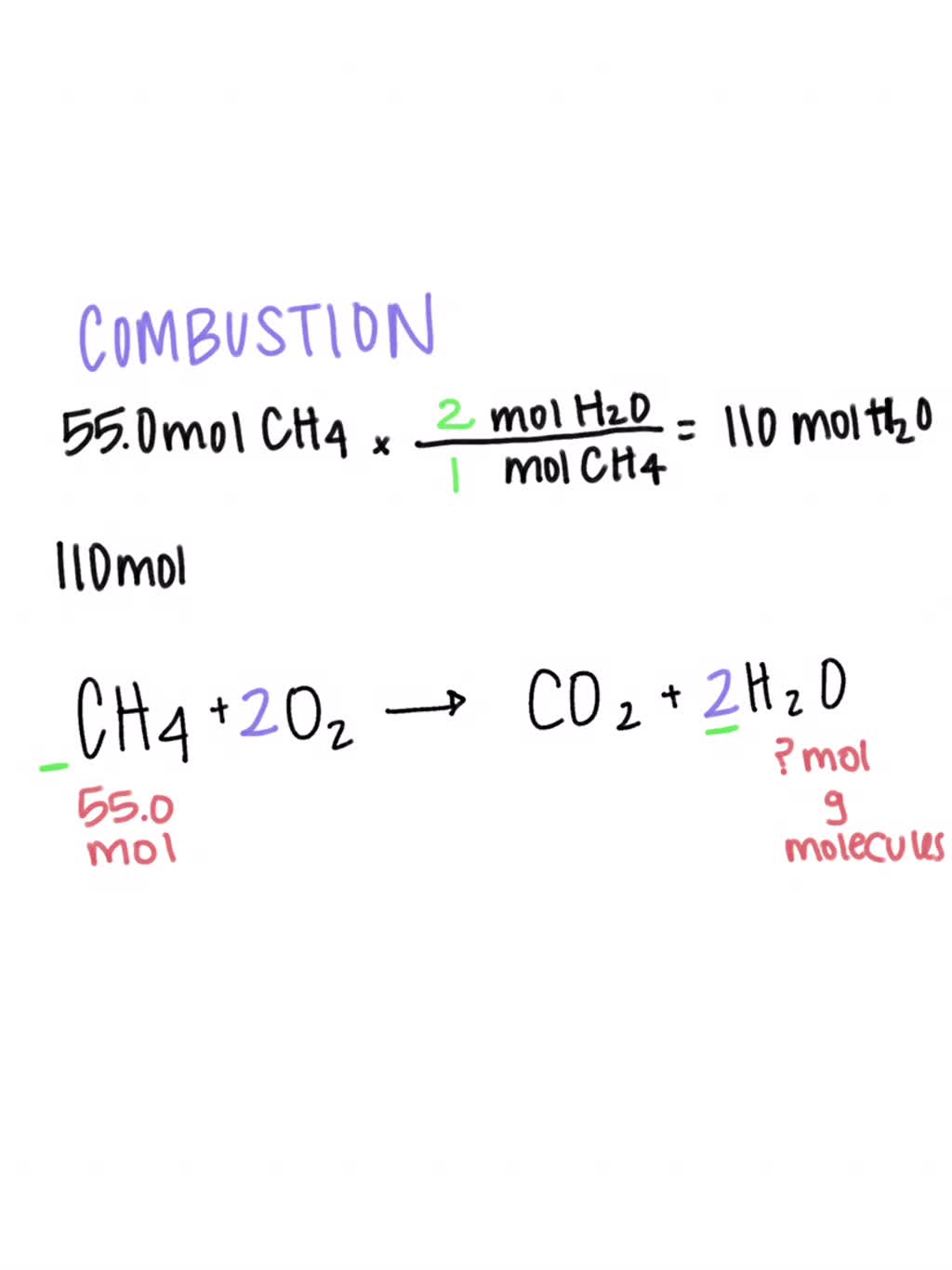
Reaction paths flux analysis for O2, CO, H2, H2O and CO2 in a He/CH4/O2... | Download Scientific Diagram

Impact of oxidants O2, H2O, and CO2 on graphene oxidation: A critical comparison of reaction kinetics and gasification behavior - ScienceDirect

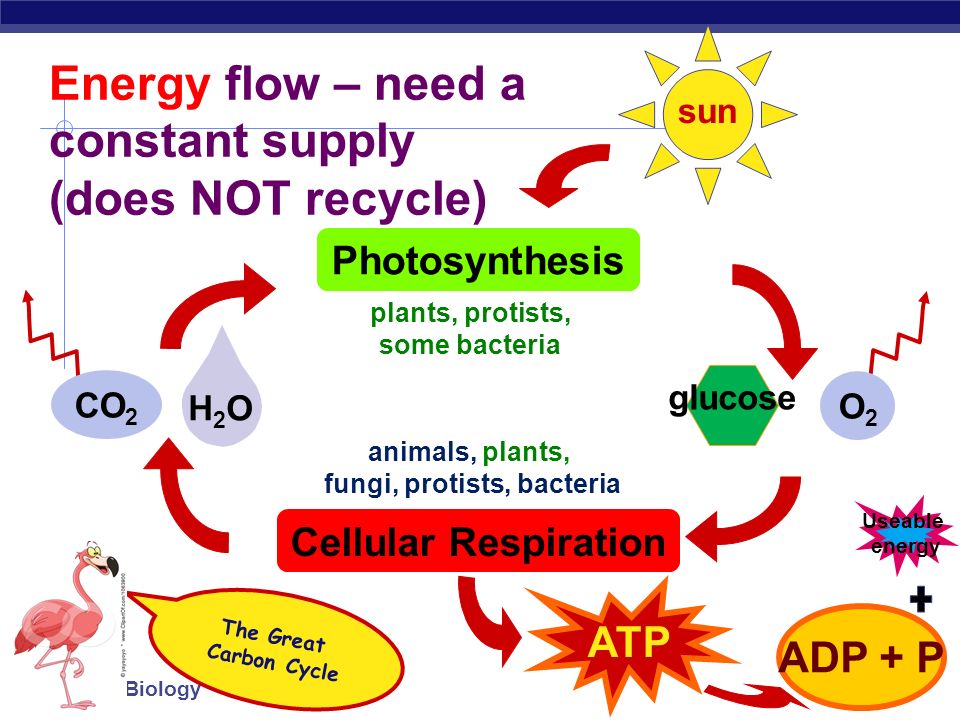
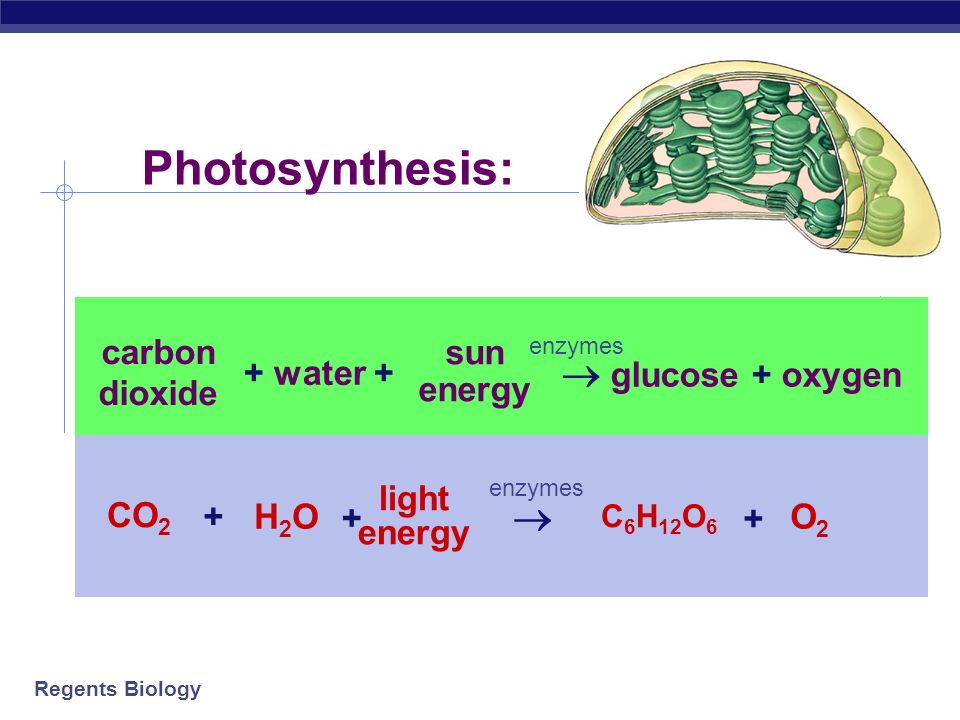
CO2+H2O=C6H12O6+O2+H2O balance the chemical equation @mydocumentary838. co2+ h2o=c6h12o6+o2+h2o - YouTube

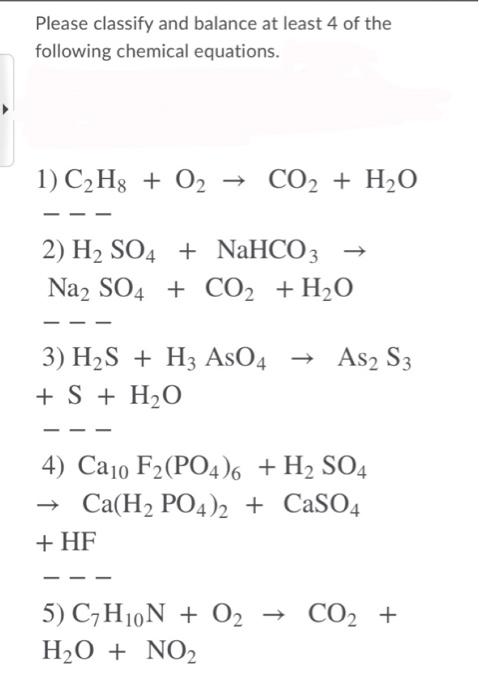
C6H6 +O2 ------}CO2 + H2O balance By oxi Methods - Chemistry - Redox Reactions - 13233357 | Meritnation.com