Consider the following reaction: H2O(l)→H2O(g);ΔH1 = 44kJ 2CH3OH(l) + 3O2→ 4H2O(l) + 2CO2(g) ; ΔH2 = - 1453kJ What is the value of Δ H for the second reaction if water vapor

See: Calculate the amount of heat released when 27.0 g H2O is cooled from a liquid at 314 K to a solid at - Brainly.com
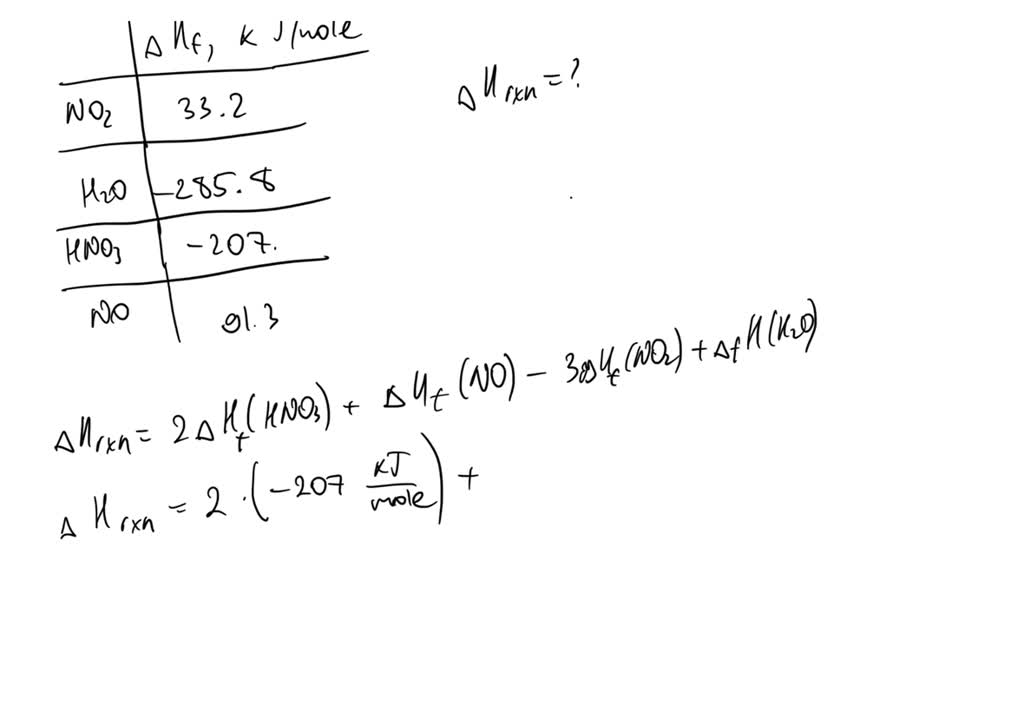
SOLVED: 4.) Consider the following reaction: 3NO2 (g) + H2O (l) ———–> 2HNO3 (aq) + NO (g) given the standard enthalpy of formations: NO2 (g) = 33.2 kJ/mole , H2O (l) = -