Ammonia Catalyzed Formation of Sulfuric Acid in Troposphere: The Curious Case of a Base Promoting Acid Rain | The Journal of Physical Chemistry A
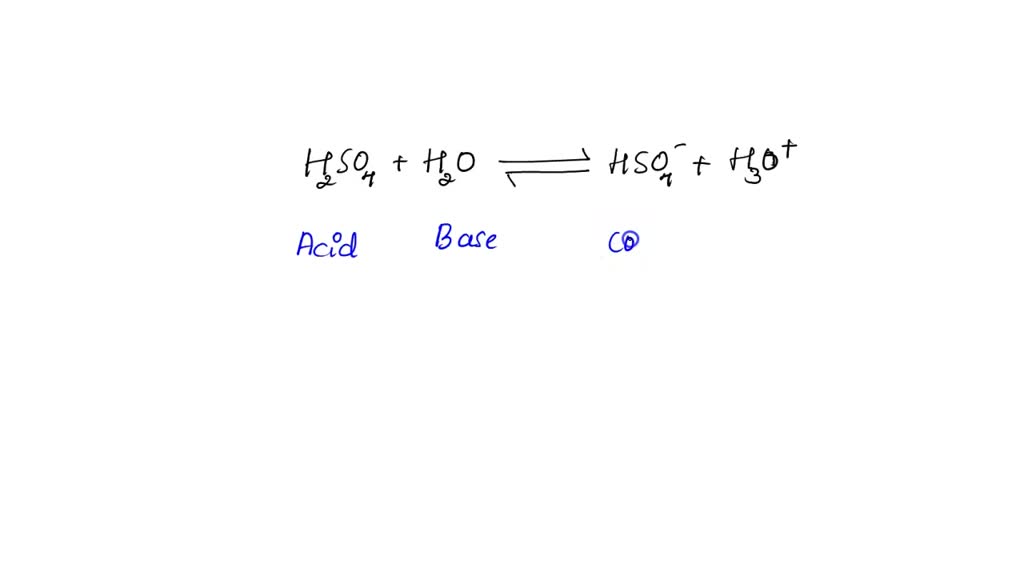
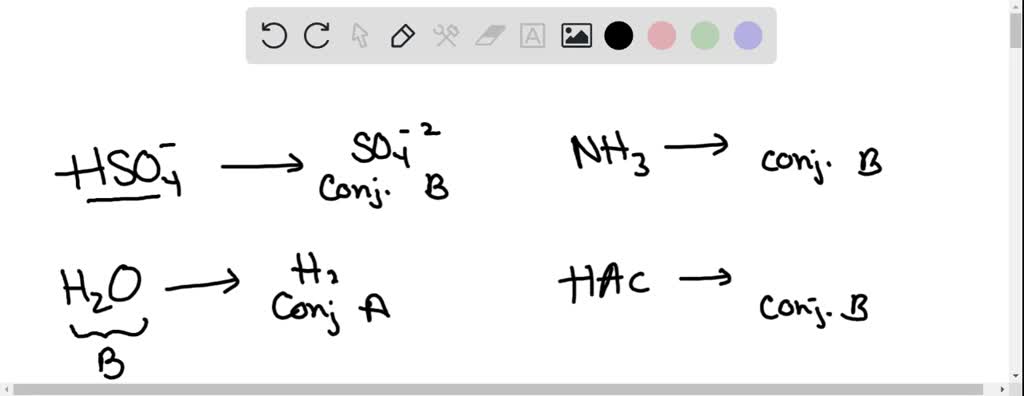
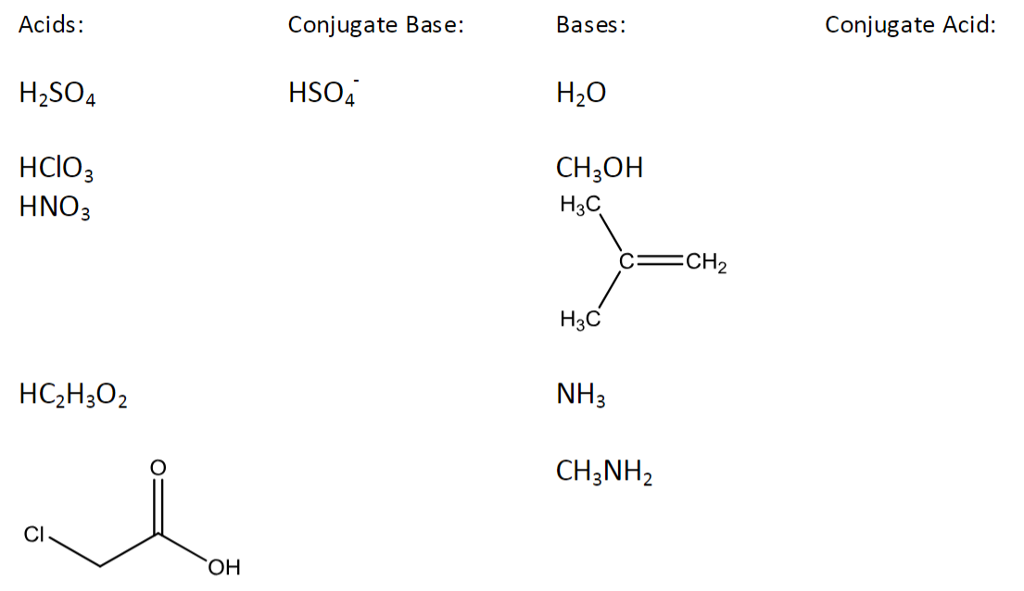
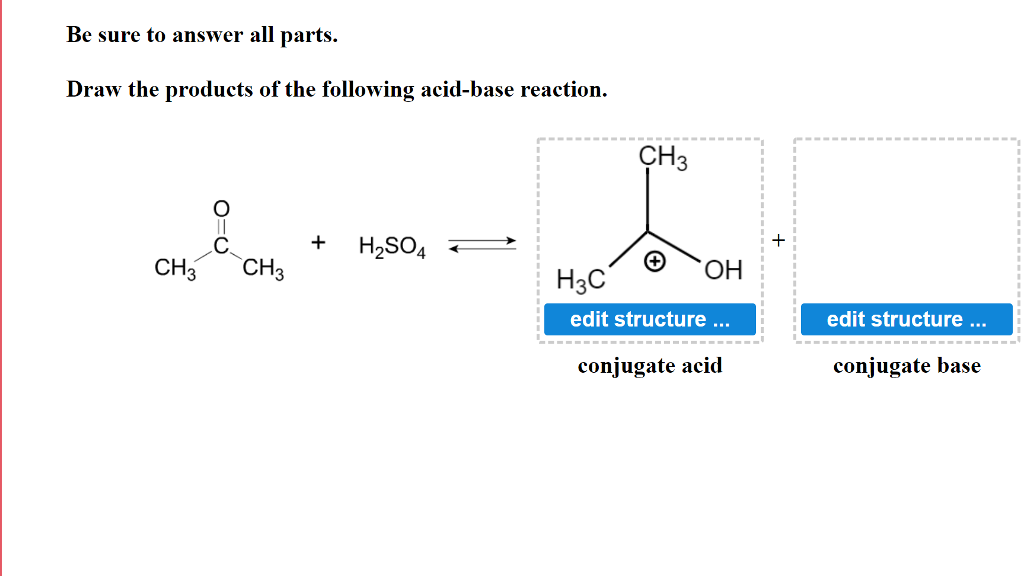
SOLVED: In the following equation, identify the Bronsted Lowry conjugate base H2SO4 + H2O <-> HSO4 - + H3O+ Group of answer choices H2SO4 HSO4- H2O H3O+

Question Video: Calculating the Volume of Sulfuric Acid That Completely Neutralizes a Given Volume and Concentration of Sodium Hydroxide | Nagwa