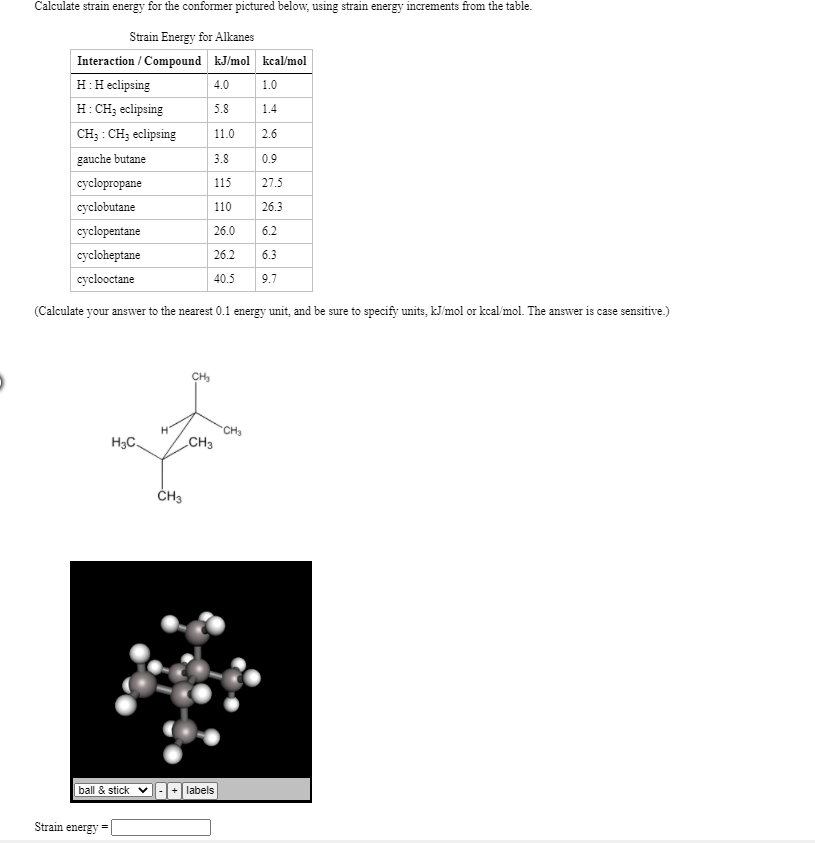
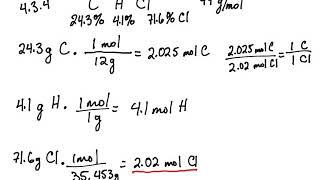If △ H(c h)= 300 kJ/mol, △ H(atm) of C(s)= 500 kJ and △ H(h h) = 200 kJ/mol then the △ fH(CH4)will be

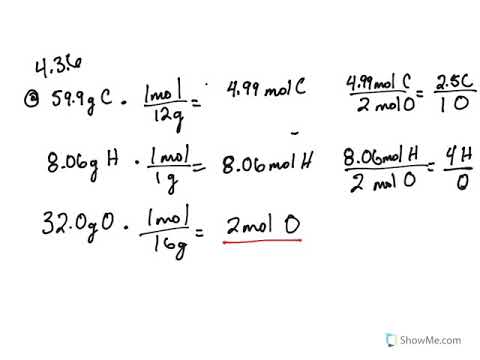
SMC: Chem 11: Week 4, Chapter 3 Combustion Analysis: 91*, 92*, 94*, 138* -Scholefield Flashcards | Quizlet

SOLVED: 1 mol of hydrocarbon P (contains C and H only) required 240 g of oxygen (O2) for complete combustion at standard state heat of combustion of P(l) is -3350 kJ/mol while

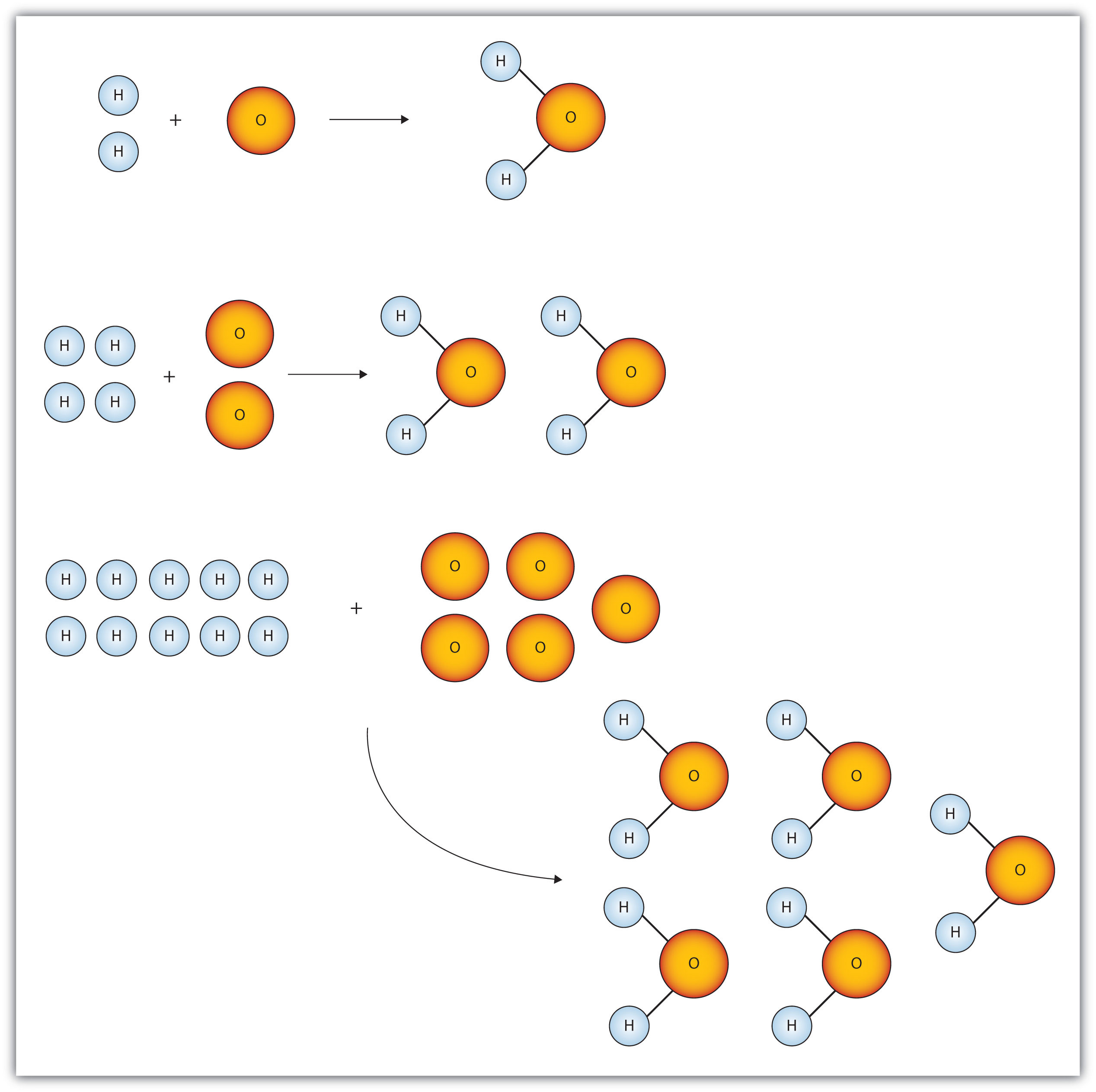
Mol ratio: coefficients of a balanced equation 2 H 2 + O 2 → 2 H 2 O 2 mol H 2 for every 1 mol O 2 In chemical calculations, mol ratios convert moles of. - ppt download