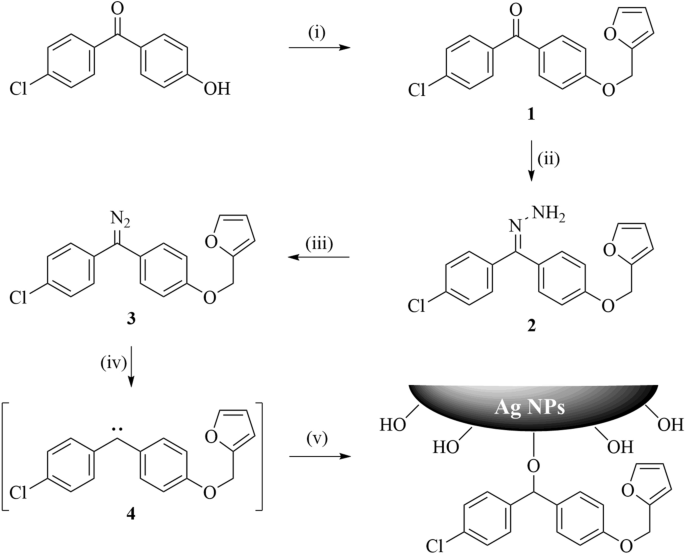
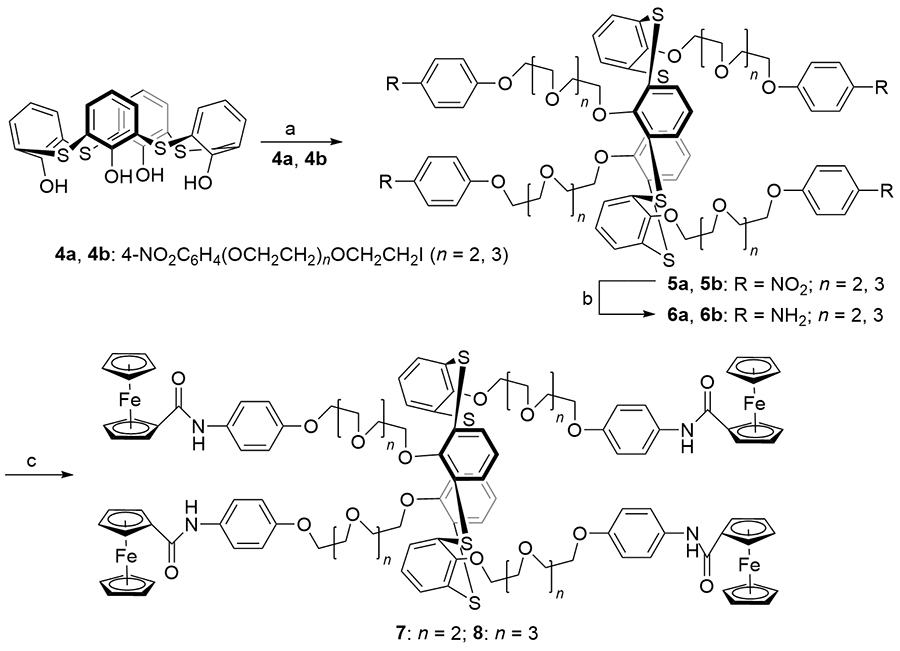
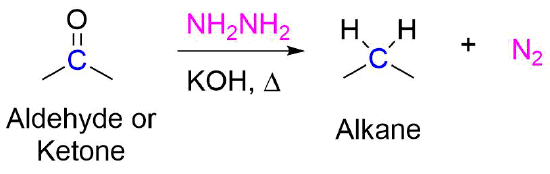Scheme 1 | Synthesis, Urease Inhibition, Antioxidant, Antibacterial, and Molecular Docking Studies of 1,3,4-Oxadiazole Derivatives

Figure 1 from Synthesis and characterization of 1,2,4-triazole containing hydrazide-hydrazones derived from (S)-naproxen as anticancer agents | Semantic Scholar
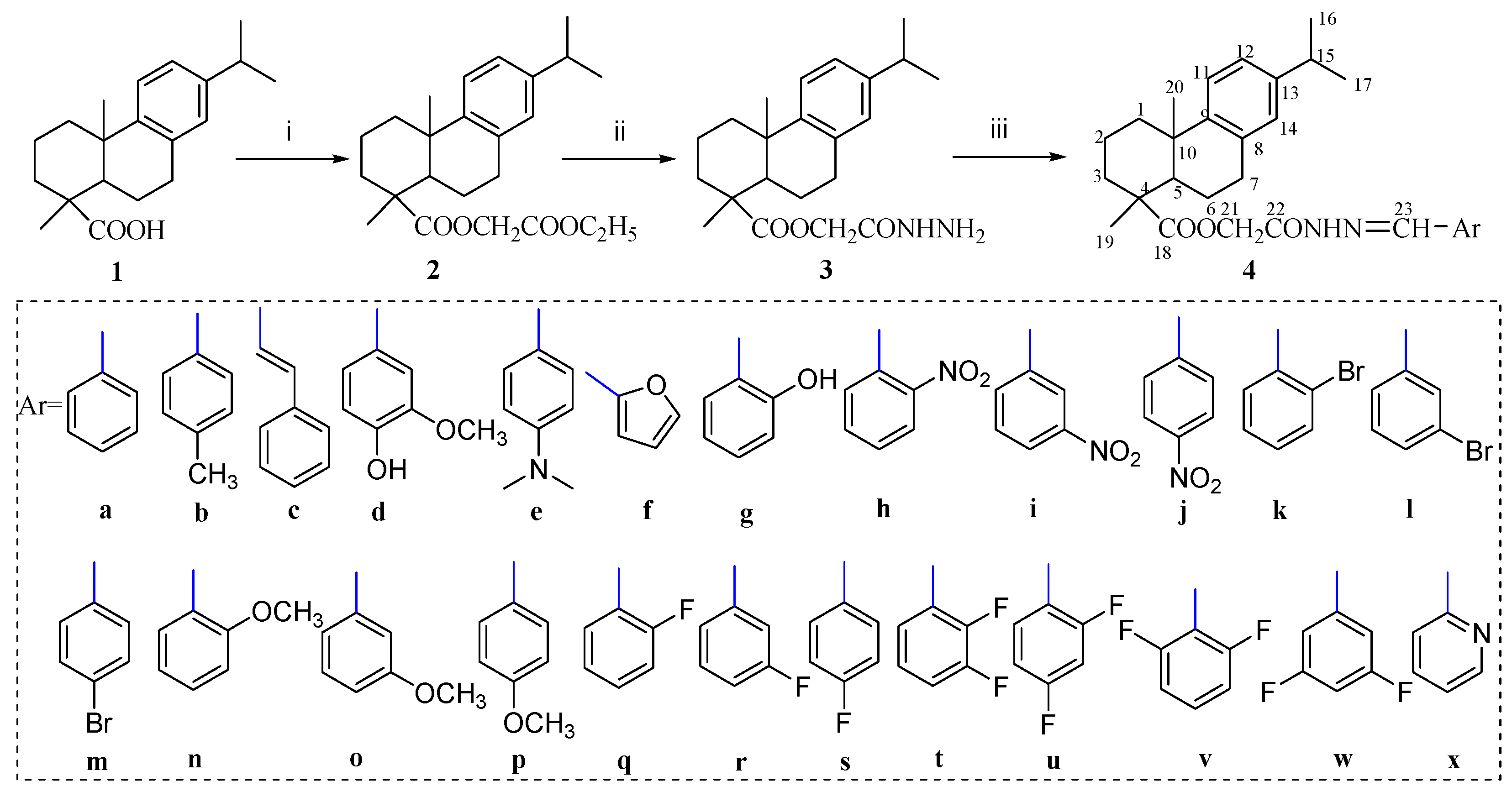
Molecules | Free Full-Text | Synthesis and In Vitro Anticancer Activity of Novel Dehydroabietic Acid-Based Acylhydrazones