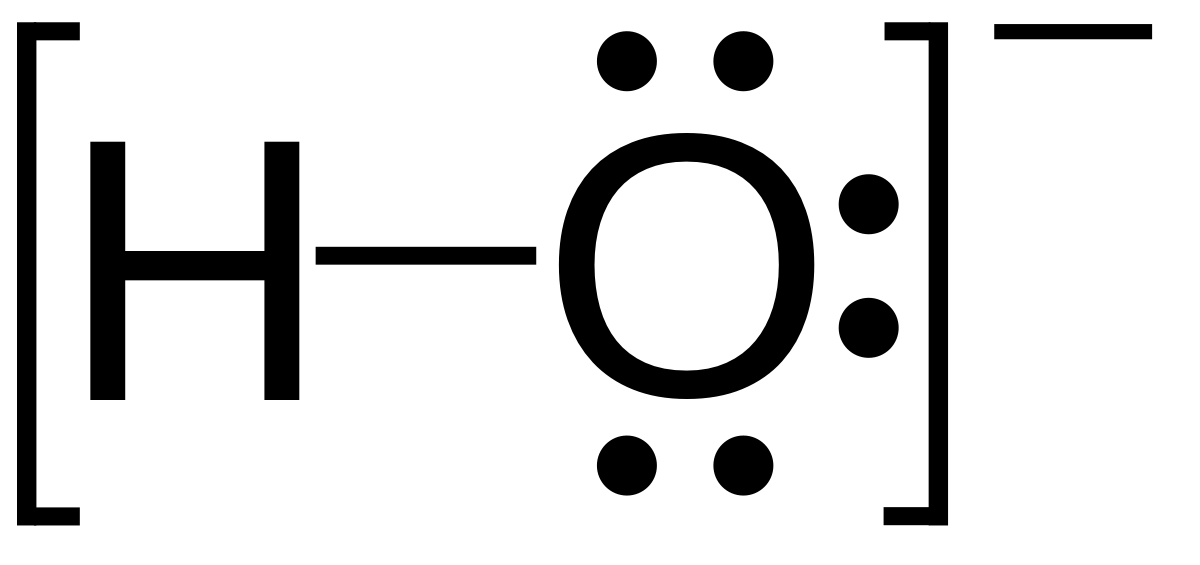
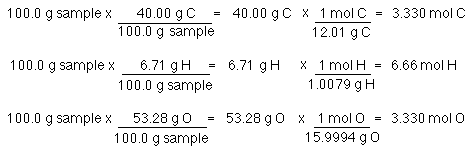Moles”. “Moles” MOLE is a way of translating between grams and amu's a mole is a number that helps us translate between the atomic world and our. - ppt download
What is the molecular formula of a compound with a percent composition of 49.47% C, 5.201% H, 28.84% N, and 16.48% O, and a molecular mass of 194.2 g/ mol? - Quora

Chemical structure of Astaxanthin, Molecular formula -C 40 H 52 O 4 and... | Download Scientific Diagram

Energy level diagram for the molecular orbitals of OH ). H and O atom... | Download Scientific Diagram

SOLVED: Determine the number of moles of oxygen atoms in each sample. a. 4.88 mol H2O2 b. 2.15 mol N2O c. 0.0237 mol H2CO3 d. 24.1 mol CO2