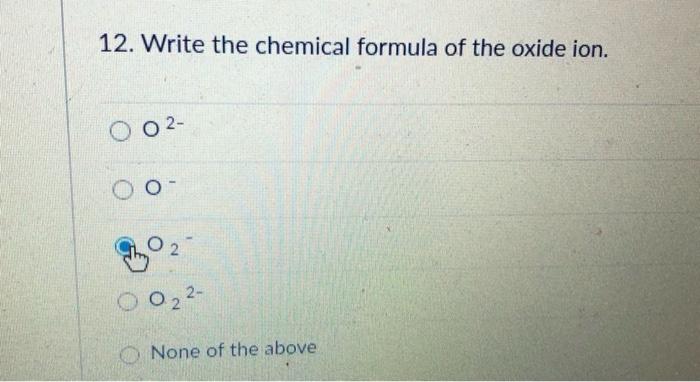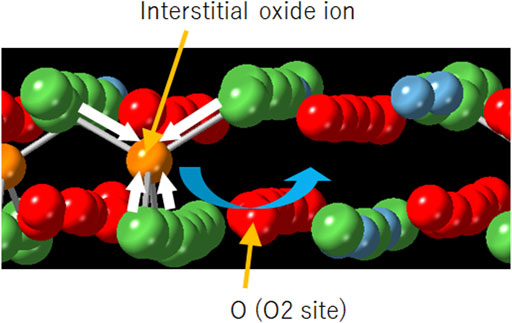
Frontiers | Effects of Ca substitution on the local structure and oxide–ion behavior of layered perovskite lanthanum nickelate
How to find whether an atom is oxide, superoxide or peroxide and all the possible types of relationships between these 3 - Quora
![PDF] Catalytic oxidation of ammonium ion in water with ozone over metal oxide catalysts | Semantic Scholar PDF] Catalytic oxidation of ammonium ion in water with ozone over metal oxide catalysts | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/df8759dfe4fdeed2bfb9e7db78f3cf68267986c4/23-Table1-1.png)
PDF] Catalytic oxidation of ammonium ion in water with ozone over metal oxide catalysts | Semantic Scholar

For the phosphorus (V) tetra oxide ion, draw the Lewis structures for all the most probable resonance structures and assign formal charges next to all atoms. | Homework.Study.com

Li2CO3 decomposition in Li-ion batteries induced by the electrochemical oxidation of the electrolyte and of electrolyte impurities - ScienceDirect




![Oxidation • einfach erklärt, Definition, Beispiele · [mit Video] Oxidation • einfach erklärt, Definition, Beispiele · [mit Video]](https://d1g9li960vagp7.cloudfront.net/wp-content/uploads/2021/03/WP-Bild_Ethanol-Oxidation-2-1024x576.jpg)